3D Printed Microscopy Chambers: Difference between revisions
mNo edit summary |
No edit summary |
||
| Line 3: | Line 3: | ||
In addition to directly printing chambers, 3D printing can also be used to make moulds for casting PDMS chambers, as described in our publication in Biochemistry and Cell Biology<ref name=":0" />. | In addition to directly printing chambers, 3D printing can also be used to make moulds for casting PDMS chambers, as described in our publication in Biochemistry and Cell Biology<ref name=":0" />. | ||
==Chamber Designs== | |||
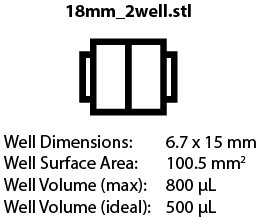
Chambers are available to fit 18 mm x 18 mm and 24 mm x 50 mm coverslips, with different well arrangements. We are still testing some of these designs, but the images below will link to the corresponding .STL file once designs are finalzed. | |||
{| class="wikitable" | {| class="wikitable" | ||
|+ | |+ | ||
| Line 39: | Line 40: | ||
|} | |} | ||
== FDM (Filament) Printing == | |||
FDM printing is the easiest in terms of printing and post-printing assembly, but it is more difficult to get a good seal with the coverslip than with DLP (resin) printing. It is important to use a good quality filament free of potentially cytotoxic or fluorescent compounds - we recommend a black or unpigmented, FDA-food grade certified PLA or PETG filament such as those sold by [https://filaments.ca/pages/search-results-page?q=fda Filaments.ca]. | |||
[[File:Assembly.png|thumb|When printing, be certain that the coverslip-contacting part of the print faces upwards.]] | |||
=== Recommended Print Settings: === | |||
* 0.4 mm diameter nozzle | |||
* 100% infill | |||
* 0.1 mm layer height | |||
* Print speed/acceleration set for high quality prints (varies with printer) | |||
* Ironing: | |||
** Top-most surface for chambers (this helps with the bond to the coverslip) | |||
** None on Lids | |||
** All top surfaces for alignment guides | |||
* No supports | |||
=== FDM Printing === | |||
# Layout your desired prints using PrusaSlicer (or your preferred slicing software). Be certain to position the prints such that the surface that will contact the coverslip face upwards. The numbers and letters on the chamber will be upside-down if the chamber is positioned correctly. | |||
# Configure the print settings as above and slice. | |||
# Print | |||
# Carefully remove the print from the print bed and wash for 15 min in distilled water. | |||
# Dry chamber before assembly (see below). | |||
== DLP (Resin) Printing == | |||
[[File:Resin.png|thumb|Only resin currently validated for cell-contact use.]] | |||
DLP (resin) printing provides superior resolution and a flatter surface for bonding the coverslip to the chamber. However, the prints must be treated carefully to remove any residual polymer or photocatalyst, as these are highly cytotoxic. At this time the only verified resin for this purpose is [https://www.amazon.ca/s?k=eSun+eResin-PLA+Bio-Photopolymer+Resin+Black&i=industrial&crid=L2CCXTG76DPP&sprefix=esun+eresin-pla+bio-photopolymer+resin+black%2Cindustrial%2C62&ref=nb_sb_noss eSun's eResin-PLA Bio-Photopolymer Resin (Black)]. | |||
=== Recommended Print Settings: === | |||
* Maximum x/y resolution | |||
* No anti-aliasing | |||
* 0.05 mm layer height | |||
* Exposure times need to be optimized for individual instruments, but for our Elegoo Mars 3: | |||
** 4 burn-in layers @ 60 s | |||
*** 0 transition layers | |||
*** 4 s light-off delay | |||
*** 6 mm lift | |||
*** 0.4 mm/m lift speed | |||
*** 150 mm/m retract speed | |||
** Normal layers: | |||
*** 0.05 mm layer height | |||
*** 8.5 s exposure | |||
*** 0 mm lift distance | |||
*** 65 mm/m lift speed | |||
*** 150 mm/m retract speed | |||
*** 0 light-off delay | |||
[[File:ResinPrint.png|thumb|Correct orientation, rotation, & supports]] | |||
=== DLP Printing: === | |||
# Layout prints as desired in Lychee Slicer or Chitubox (or your preferred slicing software). Be certain to position the prints such that the surface that will contact the coverslip face upwards. The numbers and letters on the chamber will be upside-down if the chamber is positioned correctly. | |||
# Rotate the prints 40° on the short axis (e.g. objects are rotated along the long axis, image to right). | |||
== References == | |||
<references /> | <references /> | ||
Revision as of 18:03, 2 March 2022
Customized imaging chambers can be 3D printed using FDM or SLA printing technology, and attached to a #1.5 thickness coverglass to allow for high-resolution imaging. When prepared properly, these chambers show no cytotoxicity or other signs of altered cell behaviour, even in cells cultured for multiple days. These are the chamber designs used in the Heit Lab, and are based on our publication in Biochemistry and Cell Biology[1], and on the Mod3D preprint[2] by the Truant Lab.
In addition to directly printing chambers, 3D printing can also be used to make moulds for casting PDMS chambers, as described in our publication in Biochemistry and Cell Biology[1].
Chamber Designs
Chambers are available to fit 18 mm x 18 mm and 24 mm x 50 mm coverslips, with different well arrangements. We are still testing some of these designs, but the images below will link to the corresponding .STL file once designs are finalzed.
 |
 |
 |
|---|---|---|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
FDM (Filament) Printing
FDM printing is the easiest in terms of printing and post-printing assembly, but it is more difficult to get a good seal with the coverslip than with DLP (resin) printing. It is important to use a good quality filament free of potentially cytotoxic or fluorescent compounds - we recommend a black or unpigmented, FDA-food grade certified PLA or PETG filament such as those sold by Filaments.ca.

Recommended Print Settings:
- 0.4 mm diameter nozzle
- 100% infill
- 0.1 mm layer height
- Print speed/acceleration set for high quality prints (varies with printer)
- Ironing:
- Top-most surface for chambers (this helps with the bond to the coverslip)
- None on Lids
- All top surfaces for alignment guides
- No supports
FDM Printing
- Layout your desired prints using PrusaSlicer (or your preferred slicing software). Be certain to position the prints such that the surface that will contact the coverslip face upwards. The numbers and letters on the chamber will be upside-down if the chamber is positioned correctly.
- Configure the print settings as above and slice.
- Carefully remove the print from the print bed and wash for 15 min in distilled water.
- Dry chamber before assembly (see below).
DLP (Resin) Printing

DLP (resin) printing provides superior resolution and a flatter surface for bonding the coverslip to the chamber. However, the prints must be treated carefully to remove any residual polymer or photocatalyst, as these are highly cytotoxic. At this time the only verified resin for this purpose is eSun's eResin-PLA Bio-Photopolymer Resin (Black).
Recommended Print Settings:
- Maximum x/y resolution
- No anti-aliasing
- 0.05 mm layer height
- Exposure times need to be optimized for individual instruments, but for our Elegoo Mars 3:
- 4 burn-in layers @ 60 s
- 0 transition layers
- 4 s light-off delay
- 6 mm lift
- 0.4 mm/m lift speed
- 150 mm/m retract speed
- Normal layers:
- 0.05 mm layer height
- 8.5 s exposure
- 0 mm lift distance
- 65 mm/m lift speed
- 150 mm/m retract speed
- 0 light-off delay
- 4 burn-in layers @ 60 s

DLP Printing:
- Layout prints as desired in Lychee Slicer or Chitubox (or your preferred slicing software). Be certain to position the prints such that the surface that will contact the coverslip face upwards. The numbers and letters on the chamber will be upside-down if the chamber is positioned correctly.
- Rotate the prints 40° on the short axis (e.g. objects are rotated along the long axis, image to right).
References
- ↑ 1.0 1.1 Tepperman A, Zheng DJ, Taka MA, Vrieze A, Le Lam A, Heit B. Customizable live-cell imaging chambers for multimodal and multiplex fluorescence microscopy. Biochem Cell Biol. 2020 Oct;98(5):612-623. doi: 10.1139/bcb-2020-0064. Epub 2020 Apr 27. PMID: 32339465. Link to Paper. Link to Preprint.
- ↑ C. Barba Bazan, S. Goss, C. Peng, N. Begeja, CE. Suart, K. Neuman, Ray Truant. Mod3D: A Low-Cost, Flexible Modular System of Live-Cell Microscopy Chambers and Holders. bioRxiv 2021.10.18.462400; doi: https://doi.org/10.1101/2021.10.18.462400